Abstract
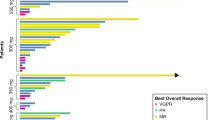
PIM447, a pan-proviral integration site for Moloney leukemia (PIM) kinase inhibitor, has shown preclinical activity in multiple myeloma (MM). This phase I, open-label, multicenter, dose-escalation study aimed to determine the maximum tolerated dose (MTD) and recommended dose for expansion (RDE) of PIM447 in Japanese patients with relapsed and/or refractory (R/R) MM. The study included 13 patients (250 mg once daily (QD), [n = 7]; 300 mg QD, [n = 6]). The sole dose-limiting toxicity observed was grade 3 QTc prolongation in one patient from the 300 mg group, and the MTD and RDE was not determined. The most common suspected PIM447-related adverse events (AEs) included thrombocytopenia (76.9%), anemia (53.8%), and leukopenia (53.8%). All patients experienced at least one grade 3 or 4 AE, most frequently thrombocytopenia or leukopenia (61.5% each). The overall response rate was 15.4%, disease control rate 69.2%, clinical benefit rate 23.1%, and two patients had a partial response (one in each dose group). Two patients treated with 250 mg QD had a progression-free survival > 6 months. PIM447 250 mg or 300 mg QD was tolerated in Japanese patients with R/R MM. Further studies are required to evaluate clinical outcomes of PIM447 in combination with other drugs for the treatment of MM.
Trial registration: clinicaltrials.gov: (NCT02160951).


Similar content being viewed by others
References
Rollig C, Knop S, Bornhauser M. Multiple myeloma. Lancet. 2015;385:2197–208.
Yan X, Clemens PL, Puchalski T, Lonial S, Lokhorst H, Voorhees PM, et al. Influence of disease and patient characteristics on daratumumab exposure and clinical outcomes in relapsed or refractory multiple myeloma. Clin Pharmacokinet. 2018;57:529–38.
Gengenbach L, Reinhardt H, Ihorst G, Ajayi S, Dold SM, Kohler M, et al. Navigating the changing multiple myeloma treatment landscape: clinical practice patterns of MM patients treated in- and outside German DSMM study group trials. Leuk Lymphoma. 2018;59:2692–9.
Paino T, Garcia-Gomez A, Gonzalez-Mendez L, San-Segundo L, Hernandez-Garcia S, Lopez-Iglesias AA, et al. The novel pan-PIM kinase inhibitor, PIM447, displays dual antimyeloma and bone-protective effects, and potently synergizes with current standards of care. Clin Cancer Res. 2017;23:225–38.
Brault L, Gasser C, Bracher F, Huber K, Knapp S, Schwaller J. PIM serine/threonine kinases in the pathogenesis and therapy of hematologic malignancies and solid cancers. Haematologica. 2010;95:1004–15.
Nawijn MC, Alendar A, Berns A. For better or for worse: the role of Pim oncogenes in tumorigenesis. Nat Rev Cancer. 2011;11:23–34.
Asano J, Nakano A, Oda A, Amou H, Hiasa M, Takeuchi K, et al. The serine/threonine kinase Pim-2 is a novel anti-apoptotic mediator in myeloma cells. Leukemia. 2011;25:1182–8.
Keane NA, Reidy M, Natoni A, Raab MS, O’Dwyer M. Targeting the Pim kinases in multiple myeloma. Blood Cancer J. 2015;5:e325.
Ramachandran J, Santo L, Siu KT, Panaroni C, Raje N. Pim2 is important for regulating DNA damage response in multiple myeloma cells. Blood Cancer J. 2016;6:e462.
Hiasa M, Teramachi J, Oda A, Amachi R, Harada T, Nakamura S, et al. Pim-2 kinase is an important target of treatment for tumor progression and bone loss in myeloma. Leukemia. 2015;29:207–17.
Garcia PD, Langowski JL, Holash J, Burger M, Zang R, Zavorotinskaya T, et al. The pan-PIM kinase inhibitor LGH447 shows activity in PIM2-dependent multiple myeloma and in AML models. Blood. 2013;122(21):1666 ((Abstract 1666)).
Nair JR, Caserta J, Belko K, Howell T, Fetterly G, Baldino C, et al. Novel inhibition of PIM2 kinase has significant anti-tumor efficacy in multiple myeloma. Leukemia. 2017;31:1715–26.
Raab MS, Thomas SK, Ocio EM, Guenther A, Goh YT, Talpaz M, et al. The first-in-human study of the pan-PIM kinase inhibitor PIM447 in patients with relapsed and/or refractory multiple myeloma. Leukemia. 2019;33:2924–33.
Babb J, Rogatko A, Zacks S. Cancer phase I clinical trials: efficient dose escalation with overdose control. Stat Med. 1998;17:1103–20.
Neuenschwander B, Branson M, Gsponer T. Critical aspects of the Bayesian approach to phase I cancer trials. Stat Med. 2008;27:2420–39.
Durie BG, Harousseau JL, Miguel JS, Blade J, Barlogie B, Anderson K, et al. International uniform response criteria for multiple myeloma. Leukemia. 2006;20:1467–73.
Raab MS, Ocio EM, Thomas SK, Günther A, Goh YT, Lebovic D, et al. Phase 1 study update of the novel pan-PIM kinase inhibitor LGH447 in patients with relapsed/refractory multiple myeloma. Blood. 2014;124(21):301 ((Abstract 301)).
An N, Kraft AS, Kang Y. Abnormal hematopoietic phenotypes in Pim kinase triple knockout mice. J Hematol Oncol. 2013;6:12.
Garcia PD, Langowski JL, Wang Y, Chen M, Castillo J, Fanton C, et al. Pan-PIM kinase inhibition provides a novel therapy for treating hematologic cancers. Clin Cancer Res. 2014;20:1834–45.
Lu J, Zavorotinskaya T, Dai Y, Niu XH, Castillo J, Sim J, et al. Pim2 is required for maintaining multiple myeloma cell growth through modulating TSC2 phosphorylation. Blood. 2013;122:1610–20.
Paíno T, San-Segundo L, Hernández-García S, González-Méndez L, Algarin EM, Martín-Sánchez M et al. The pan-PIM kinase inhibitor, PIM447, potentially synergizes with pomalidomide plus dexamethasone in preclinical in vitro and in vivo models of multiple myeloma. EHA Library. 2017;(180998):E1222. (Abstract E1222).
Acknowledgments
The study was supported by Novartis Pharmaceuticals Corporation. We thank the investigators of the study. We thank Kazuto Natsume (previously Novartis employee) who is currently working at AbbVie GK, Norifumi Ishikawa (previously Novartis employee) currently working at Nobelpharma Co., Ltd and K. Gary Vanasse (previously Novartis employee) for their significant contribution to this study. We also thank Bhavani Yamsani, M. Pharm, MBA, and Ambrin Fatima, PhD, of Novartis Healthcare Pvt. Ltd. for providing medical editorial and writing assistance with this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Iida, S., Sunami, K., Minami, H. et al. A phase I, dose-escalation study of oral PIM447 in Japanese patients with relapsed and/or refractory multiple myeloma. Int J Hematol 113, 797–806 (2021). https://doi.org/10.1007/s12185-021-03096-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-021-03096-9